Allosteric inhibition of SHP2 uncovers aberrant TLR7 trafficking in aggravating psoriasis
Yuyu Zhu,Zhigui Wu#,Wei Yan,Fenli Shao,Bowen Ke,Xian Jiang,Jian Gao,Wenjie Guo,Yuping Lai,Hongyue Ma,Dijun Chen,Qiang Xu,Yang Sun
EMBO Mol Med. 2022 Mar 7; 14(3): e14455. doi: 10.15252/emmm.202114455
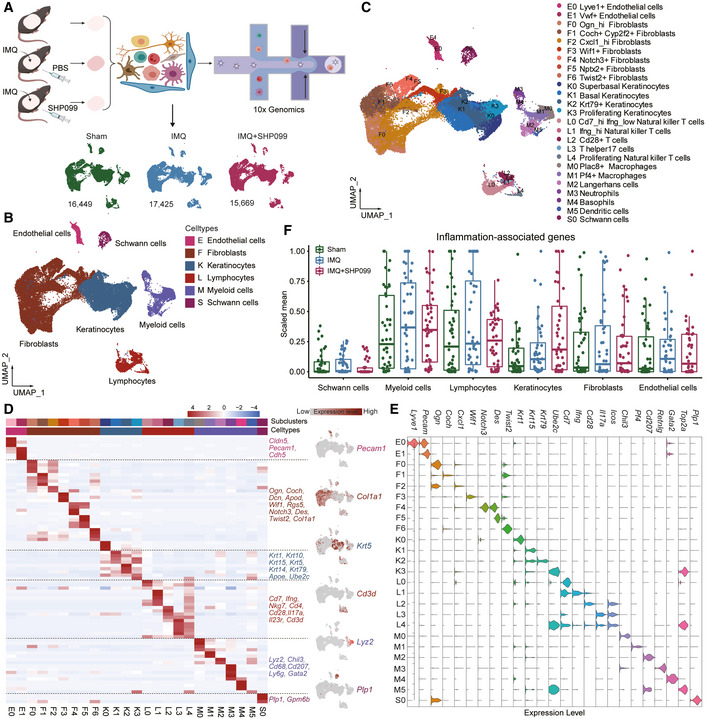
Psoriasis is a complex chronic inflammatory skin disease with unclear molecular mechanisms. We found that the Src homology-2 domain-containing protein tyrosine phosphatase-2 (SHP2) was highly expressed in both psoriatic patients and imiquimod (IMQ)-induced psoriasis-like mice. Also, the SHP2 allosteric inhibitor SHP099 reduced pro-inflammatory cytokine expression in PBMCs taken from psoriatic patients. Consistently, SHP099 significantly ameliorated IMQ-triggered skin inflammation in mice. Single-cell RNA sequencing of murine skin demonstrated that SHP2 inhibition impaired skin inflammation in myeloid cells, especially macrophages. Furthermore, IMQ-induced psoriasis-like skin inflammation was significantly alleviated in myeloid cells (monocytes, mature macrophages, and granulocytes)-but not dendritic cells conditional SHP2 knockout mice. Mechanistically, SHP2 promoted the trafficking of toll-like receptor 7 (TLR7) from the Golgi to the endosome in macrophages by dephosphorylating TLR7 at Tyr1024, boosting the ubiquitination of TLR7 and NF-魏B-mediated skin inflammation. Importantly, Tlr7 point-mutant knock-in mice showed an attenuated psoriasis-like phenotype compared to wild-type littermates following IMQ treatment. Collectively, our findings identify SHP2 as a novel regulator of psoriasis and suggest that SHP2 inhibition may be a promising therapeutic approach for psoriatic patients.