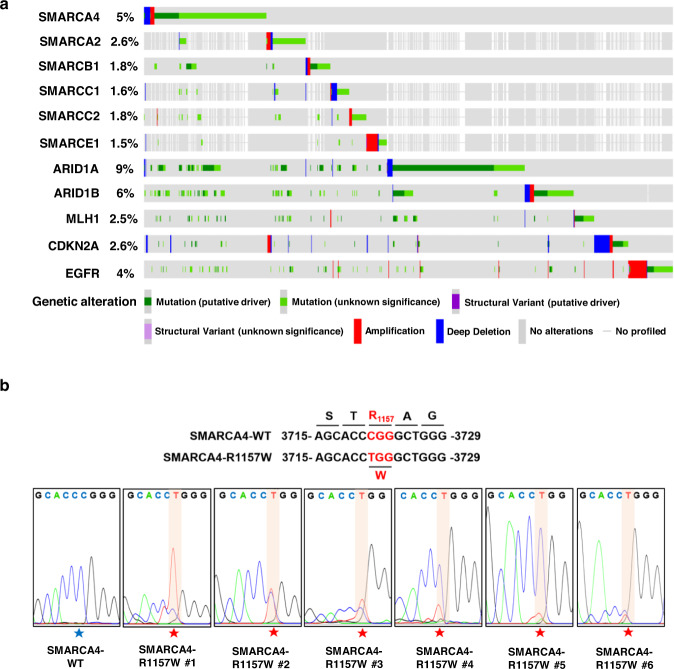
The SMARCA4(R1157W) mutation facilitates chromatin remodeling and confersPRMT1/SMARCA4 inhibitors sensitivity in colorectal cancer.
Zeng X, Yao B, Liu J, Gong GW, Liu M, Li J, Pan HF, Li Q, Yang D, Lu P, Wu D, Xu P, Chen B, Chen P,Zhang M, Zen K, Jing J, Huang DCS, Chen D, Jiang ZW, Zhao Q
NPJ Precis Oncol. 2023 Mar 15;7(1):28. doi: 10.1038/s41698-023-00367-y.
Genomic studies have demonstrated a high frequency of genetic alterations incomponents of the SWI/SNF complex including the core subunit SMARCA4. However,the mechanisms of tumorigenesis driven by SMARCA4 mutations, particularly incolorectal cancer (CRC), remain largely unknown. In this study, we identified aspecific, hotspot mutation in SMARCA4 (c. 3721C>T) which results in a conversionfrom arginine to tryptophan at residue 1157 (R1157W) in human CRC tissuesassociated with higher-grade tumors and controls CRC progression.Mechanistically, we found that the SMARCA4R1157W mutation facilitated itsrecruitment to PRMT1-mediated H4R3me2a (asymmetric dimethylation of Arg 3 inhistone H4) and enhanced the ATPase activity of SWI/SNF complex to remodelchromatin in CRC cells. We further showed that the SMARCA4R1157W mutantreinforced the transcriptional expression of EGFR and TNS4 to promote theproliferation of CRC cells and patient-derived tumor organoids. Importantly, wedemonstrated that SMARCA4R1157W CRC cells and mutant cell-derived xenograftswere more sensitive to the combined inhibition of PRMT1 and SMARCA4 which actsynergistically to suppress cell proliferation. Together, our findings show thatSMARCA4-R1157W is a critical activating mutation, which accelerates CRCprogression through facilitating chromatin recruitment and remodeling. Ourresults suggest a potential precision therapeutic strategy for the treatment ofCRC patients carrying the SMARCA4R1157W mutation.